
What Does it take to make a fire?
Fire occurs whenever combustible fuel in the presence of oxygen at an extremely high temperature becomes gas. Flames are the visual indicator of the heated gas. Once a fire is started, it must maintain optimal levels of each of these elements to sustain a flame. This is often represented as The Fire Triangle
The Fire Triangle
As children we are often taught about fire in terms of The Fire Triangle. Also known as The Combustion Triangle, we use it to understand the chemical reaction needed to create and fuel a fire and how to prevent the outbreak and spread of that fire. Using just three elements, The Fire Triangle is a clear and simple method of raising awareness of how and why fire is able to ignite and spread, making it a great tool to save lives.
Elements of The Fire Triangle
The sides of The Fire Triangle represent the interdependent ingredients needed for fire: heat, fuel and oxygen. All three elements of the triangle must be present in order to ignite a fire.
Heat
A heat source is responsible for the initial ignition of fire, and is also needed to maintain the fire and enable it to spread. How much heat is required for the chemical reaction depends on the fuel’s specific flash point—the lowest temperature at which the fuel will ignite. As a combustible material succumbs to a fire, more heat is naturally produced, allowing fire to spread by drying out and heating nearby fuel sources and warming surrounding air.
Fuel
A fire cannot ignite without an object or material present to enable the fire to burn. Fuels can be flammable solids, liquids, or gases. Technically, any substance that can combust can serve as the fuel for a fire. Businesses contain a variety of different combustible materials which will help to fuel a fire, including:
– Paper
– Office Furniture
– Cooking Oil
– Cleaning Chemicals
– Gasoline
While some materials burn more rapidly than others, a fire can spread relatively easily inside a building no matter its contents.
Oxygen
At least 16 percent oxygen must be in the air for a fire to start. The air we breathe has 21 percent oxygen, making it more than enough to allow a fire to burn. Oxygen supports the chemical processes that occur during fire. When fuel burns, it reacts with oxygen from the surrounding air. This reaction releases heat and creates the combustion products gas, smoke and embers in a process known as oxidation. The more oxygen present, the more significant the acceleration of the chemical reaction taking place.
A Fourth Element?
Recently, due to further research, the chemical chain reaction was added as the fourth element to The Fire Triangle, creating The Fire Tetrahedron. Adding a fourth dimension gives a fuller picture of what causes fires to ignite and burn over time. The chemical chain reaction keeps the fire burning by providing adequate heat to sustain the fire. As long as the chemical chain reaction is sustained, the fire will grow and continue to burn. When a fire is effectively suppressed, it is because at least one element of the fire tetrahedron—the ongoing reaction—has been removed from the equation.
How to Control a Fire
Stopping combustion requires depriving the fire of one or more sides of the fire triangle, or by stopping the chemical chain reaction that sustains combustion—and that’s how standard fire extinguishing works.
Safely removing critical elements from the fire triangle
To prevent the ignition and subsequent spread of fire, it’s important that one or more elements are removed. The action you should take depends on the type of fire and which element’s removal will cause the fire to extinguish the most efficiently and safely. All fires can be extinguished by cooling, smothering, starving or by interrupting the combustion process to extinguish the fire.
Cooling – Removing Heat
To extinguish a fire by cooling, the rate at which heat energy is lost from the burning material must be increased by removing some of the heat energy. Many fires are too big for large gusts of air to effectively cool the fuel temperature, so an agent is dispersed. When a fire is extinguished with a fire suppression chemical, clean agent or water, the rate at which heat is lost from the burning material increases. Eventually, the rate at which heat is lost from the fire may be greater than the rate of heat production and the fire will die away.
Water is a good cooling agent because of its high thermal capacity and latent heat of vaporization. This, combined with the fact it is available in large quantities, makes it by far the most widely useful fire extinguishing agent. However, water fire extinguishers cannot be used on all types of fire, and so extra care should be taken on electrical or cooking fires.
Starving – Removing Fuel
In some cases, a fire can be extinguished simply by removing the fuel source. This may be accomplished in a number of ways, such as stopping the flow of liquid or gas fuels, limiting fuel by removing potential fuel from the vicinity of the fire, allowing the fire to burn until all of the fuel is consumed, removing the fire from the mass of combustible materials or by dividing the burning material into smaller fires that can be extinguished more easily.
Follow all building codes and use non flammable or fire retardant materials where required. This will help to prevent the fire from spreading and igniting non-fire-resistant materials.
Smothering – Removing Oxygen
Smothering extinguishes a fire by isolating it from the supply of oxygen. By preventing oxygen rich air flow from reaching the fire in a confined atmosphere, the chemical reaction during combustion forces the fire to consume the remaining oxygen until there is no longer enough to sustain a flame, thus the fire will be extinguished.
Fire blankets and extinguishers are designed to quickly put out fires by cutting off the fuel from the supply of oxygen. Other fire suppression agents work by lowering the oxygen concentration. For example, CO2 reduces oxygen and aids in heat removal while other inert gases are mainly lower oxygen concentration. Fire doors will also help to prevent a sufficient amount of oxygen from reaching the site of a fire.
Let A P Fire Help!
To help protect your people and establishment, A P Fire Protection is here to provide your commercial property with a wide range of fire safety equipment and services to help prevent the outbreak and spread of fire.
Your First Line of Defense – Fire Extinguishers
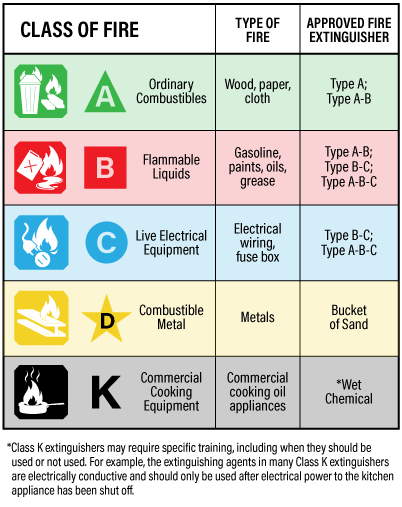
Fire extinguishers will eliminate one or more of the critical elements of a fire. Therefore, it’s important that you have the appropriate fire extinguisher on hand to deal with a particular type of fire. We have created an overview of how the most common types of agents work to disrupt the fire triangle/tetrahedron.
- Carbon dioxide (CO2) and inert gases put out Class B and C fires by removing oxygen from the fire triangle. While inert gases will also put out Class A fires, CO2 can be used for surface Class A fires only.
- Water-based systems put out Class A fires by removing heat from the fire triangle.
- Dry powders are available in a few different types, with different applications appropriate for Class A, B, or C fires, work by applying a thin layer of material to separate the fuel from surrounding oxygen, interrupting the chemical chain reaction that keeps a fire going.
- Clean agents are highly effective against Class A, B, and C fires and disrupts the fire triangle by reducing the heat or separating the oxygen.
At A P Fire Protection we are able to supply commercial premises with a wide range of fire extinguishers from Badger and Amerex, along with expert advice about where they would be best placed within your building.
Extinguishing a Fire Without a Fire Extinguisher
In certain instances, depending on the type of occupancy or equipment, it’s not always ideal to use water or a conventional fire extinguisher to put out a fire. Special circumstances like these require the use of a clean agent fire suppression system. Clean agents require no cleanup after discharge and pose no significant risk to equipment, people, or the environment. Clean agent fire suppression systems are favorable for use around equipment such as: precision machines, server rooms, electrical cabinets, data storage, and telecommunications systems, archives storing records, files or artwork, etc.
When it comes to protecting equipment, a key factor to understand is what class of fire may occur. As mentioned above, not all agents are effective for every class of fire. When selecting the best fire suppression agent and system for your situation, it is just as important to understand which class of fire you are protecting against.
In addition to fire extinguishers, we can also supply a variety of fire alarm systems, emergency lighting, fire sprinkler systems, risk assessments, and fire training. For more information about how we can help you, get in touch with our team today – we service the entire state of Arizona and soon Texas.
Whether it occurs in the workplace, home, or elsewhere, it is easy to underestimate the unique and serious dangers fire poses to people, assets, equipment, and physical structures. A burning fire is a fascinating chemical chain reaction, and extinguishing a fire is a matter of disrupting that chain reaction. The better you understand how a fire starts—including the basic science of what components are required for it to ignite and burn—the better you’ll be prepared when you need to extinguish a fire.








Leave a Reply